Monomer recovery in 86 yield involved dissolving the polymer slowly in a 05 M H 2 SO 4 solution followed by neutralizing and filtering pure monomer without further purification. Calculator ruler P62083RA P62083RA0132 2020 Pearson Education Ltd.
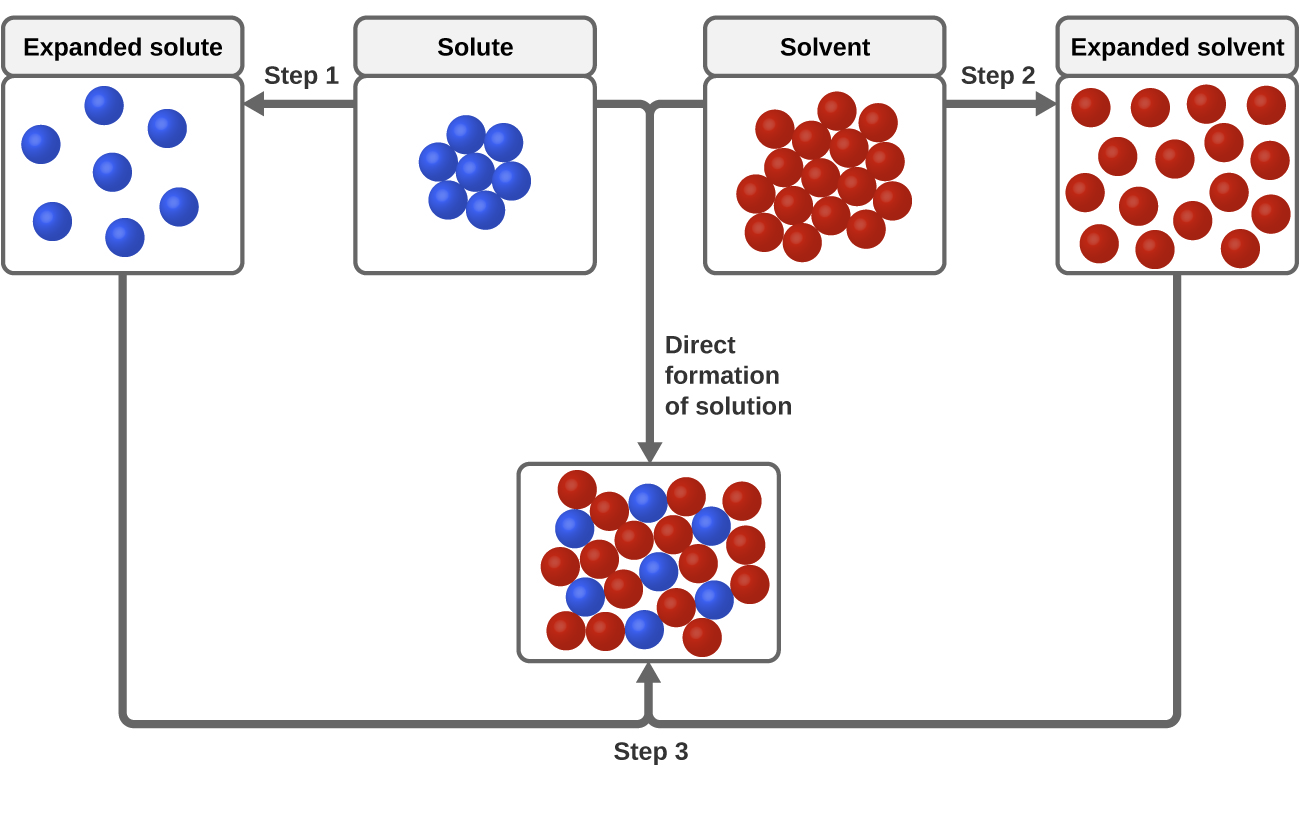
11 1 The Dissolution Process Chemistry
Activity level is often expressed as total surface area per unit weight usually in square meters per gram.

. Fill in the boxes at the top of this page with your. A Laboratory Manual Third Edition. Full PDF Package Download Full PDF Package.
Level 1Level 2 GCSE 91 Thursday 14 May 2020 Morning Time. Simple distillation fractional distillation filtration crystallisation paper chromatography. For example- if the cation is Fe 3 and the anion found is.
1 hour 45 minutes Paper Reference 1CH01F Chemistry Paper 1 Foundation Tier You must have. Download Full PDF Package. After finding the cations group take each anion of the group for performing the positivity test.
Traditionally the dissolving of the specimen is required for the standard analytical procedures for estimating the molecular weight dispersion MWD. Aside from that if the existing technology does not have a gauge for measuring precise molecular mass. Dissolving of instant coffee in water seems to be a physical change but in most cases dissolving is accompanied by an energy change and is probably better considered to be a chemical process even though it is possible to recover the original components by physical means.
Compounds with the same molecular formula but different structural formulae and determination of possible structural formulae of an organic molecule given its molecular formula R eaction mechanisms. These changes may also occur in the absence of an identifiable cause. The Interactions curriculum introduces students to science as an endeavor a process we engage in rather than solely a set of discoveries by others.
Through engaging in modeling and scientific explanation students explore curious aspects of the everyday world discovering how the unseen world of atomic level interactions and energy transformations are responsible for much of. Certain polymers on the other hand are either hard to break down or completely intractable in conventional solvents. At a critical point a stream of liquid erupts from the surface.
111 understand how a chromatogram provides information about the composition of a mixture. A Laboratory Manual Third Edition 1982. This point of eruption is known as the Taylor coneIf the molecular cohesion of the liquid is sufficiently high stream.
Many examples of materials dissolving for example an Alka Seltzer in water metal in acid and the effect of. To be useful in. For example the stimulus cannot be identified in as many as 85 of individuals.
A short summary of this paper. Separation Process Principles- Chemical and Biochemical Operations 3rd Edition. Balancing the charges of cations and anions is necessary.
When a sufficiently high voltage is applied to a liquid droplet the body of the liquid becomes charged and electrostatic repulsion counteracts the surface tension and the droplet is stretched. After obtaining and identifying both anions and cations construct the chemical formula of salt. 36 Full PDFs related to this paper.
110 describe these experimental techniques for the separation of mixtures. Empiricalmolecular formulas Mass moles and molecules relationships SC7. Changes at the molecular level are dramatic and may include alterations in genetic transcription of neurotransmitters and receptors.
Students will characterize the properties that describe solutions and the nature of acids and bases. For the blend samples 15 EDEA or HMDA molar ratio compared to FAI was added. All the samples were prepared by dissolving 5 mg FAI in 04 ml dimethyl sulfoxide-d6 DMSO-d6.
Toward the higher end of this range one might better visualize one pound about a quart in volume of granular activated carbon with a total surface area of 125 acres. 112 understand how to use the calculation of Rf values to identify the components of a mixture. One of the frustrating aspects of chronic pain is that the stimulus may be unknown.
This total exposed surface will typically be in the range of 600-1200 m2g. The same process should be followed for finding the cations. Express concentrations as molarities Relate molality to colligative properties.
111111 Turn over Instructions Use black ink or ball-point pen. Explain the process of dissolving in terms of solutesolvent interactions.

Dissolving Process Chemistry For Non Majors
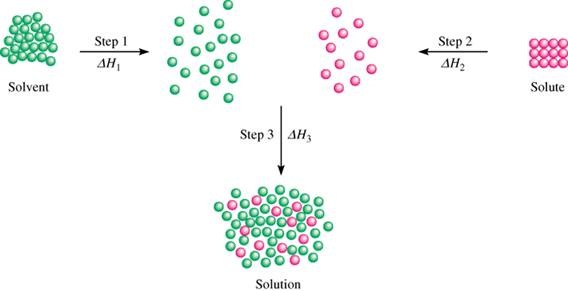
Dissolving Process Ms Jonasson S Classes

4 1c Solution Chemistry Describe The Dissolving Process Cset Study Guide Chemistry
0 Comments